Mephedrone (4-methylmethcathinone) is a semisynthetic stimulant manufactured from cathinone, the active ingredient of the African shrub khat, and has been marketed as plant food or bath salts. Cathinone is structurally very similar to amphetamine and can be considered as the β-keto analogue of amphetamine. 1 The effects on monoamine neurotransmission confirm the cathinone mechanism of action to be similar to those of amphetamines, with both groups of drugs binding to and promoting the release of monoamine transporters for dopamine, serotonin and noradrenaline. Reference Cozzi, Sievert, Shulgin, Jacobill and Ruoho2,Reference Nagai, Nonaka and Kamimura3 Mephedrone is usually sold as a white powder in capsules or zip-lock bags and is most commonly either snorted (‘insufflated’) or taken orally (‘bombed’). It costs between £10 and £30 per gram; doses are generally between 0.5 and 1 g, with some reports of binges up to 10 g in a session. Reference Newcombe4 The desirable psychoactive effects of mephedrone include euphoria, empathy, stimulation, intensification of sensory experience (e.g. music appreciation), mild sexual stimulation, mood enhancement, decreased hostility/insecurity and increased insight. 1,Reference Newcombe4
Mephedrone use in the UK grew rapidly in 2009 and early 2010, with one self-reported drug use survey finding it to be the fourth most frequently used drug (in the prior month) after cannabis, ecstasy and cocaine. Reference Dick and Torrance5 A case series from 2009 revealed agitation as the most commonly experienced sign of toxicity, occurring in 53% of the 15 individuals. Reference Wood, Greene and Dargan6
There have been a number of deaths reportedly linked with mephedrone use. 1 The UK Advisory Council on the Misuse of Drugs (ACMD) recommendation that cathinone compounds such as mephedrone be classified as class B drugs came into effect in April 2010. The UK is the first country to place a generic ban on all cathinone compounds.
Literature review revealed little data available on psychiatric symptoms and the implications for mental health associated with mephedrone use. A few cross-sectional and case-control studies found an interaction between khat use and mental disorder, Reference Warfa, Klein, Bhui, Leavey, Craig and Stansfeld7 but nothing outwith the popular press and the ACMD report on the specific psychotropic effects of mephedrone was identified.
We aimed to examine the occurrence of psychiatric symptoms due primarily to mephedrone intoxication occurring in patients presenting to Edinburgh and Falkirk health services.
Method
We identified patients presenting between January and June 2010 to the emergency departments or mental health services in Edinburgh and Falkirk with adverse effects of mephedrone use. Mephedrone ingestion was self-reported, as was other substance misuse. A questionnaire was designed to retrospectively collect case-note data, and this was applied to all identified cases. Data collected included: age; gender; previous psychiatric history; substance misuse; mephedrone use; psychiatric and physical symptoms relating to mephedrone use; and need for hospital admission. All identifying information was removed from the data.
Results
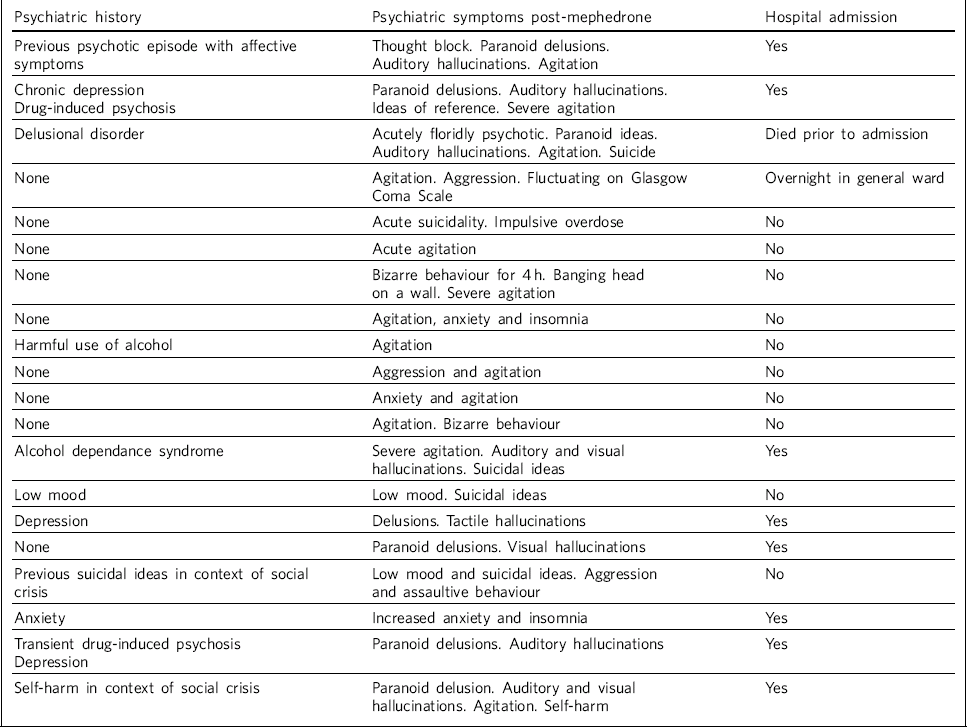
Twenty cases of mephedrone use associated with adverse psychological sequelae were ascertained from Edinburgh (n = 14) and Falkirk (n = 6) (Table 1; for full details see online Table DS1). The psychiatric symptoms noted contemporaneously ranged from agitation through to florid psychotic symptoms including auditory, visual and tactile hallucinations as well as paranoid delusions. The majority of patients (n = 17) were male, and age at time of presentation was 19-59 years, with most individuals (n = 14) falling in the 19-29 years range. Only five individuals presented with adverse effects following their first use of mephedrone, with the remaining patients reporting regular or heavy use prior to presentation. Alcohol was a complicating factor in 60% of presentations.
Table 1 Clinical details of mephedrone users self-presenting to emergency services
| Psychiatric history | Psychiatric symptoms post-mephedrone | Hospital admission |
|---|---|---|
| Previous psychotic episode with affective symptoms | Thought block. Paranoid delusions. Auditory hallucinations. Agitation | Yes |
| Chronic depression Drug-induced psychosis | Paranoid delusions. Auditory hallucinations. Ideas of reference. Severe agitation | Yes |
| Delusional disorder | Acutely floridly psychotic. Paranoid ideas. Auditory hallucinations. Agitation. Suicide | Died prior to admission |
| None | Agitation. Aggression. Fluctuating on Glasgow Coma Scale | Overnight in general ward |
| None | Acute suicidality. Impulsive overdose | No |
| None | Acute agitation | No |
| None | Bizarre behaviour for 4 h. Banging head on a wall. Severe agitation | No |
| None | Agitation, anxiety and insomnia | No |
| Harmful use of alcohol | Agitation | No |
| None | Aggression and agitation | No |
| None | Anxiety and agitation | No |
| None | Agitation. Bizarre behaviour | No |
| Alcohol dependance syndrome | Severe agitation. Auditory and visual hallucinations. Suicidal ideas | Yes |
| Low mood | Low mood. Suicidal ideas | No |
| Depression | Delusions. Tactile hallucinations | Yes |
| None | Paranoid delusions. Visual hallucinations | Yes |
| Previous suicidal ideas in context of social crisis | Low mood and suicidal ideas. Aggression and assaultive behaviour | No |
| Anxiety | Increased anxiety and insomnia | Yes |
| Transient drug-induced psychosis Depression | Paranoid delusions. Auditory hallucinations | Yes |
| Self-harm in context of social crisis | Paranoid delusion. Auditory and visual hallucinations. Agitation. Self-harm | Yes |
The most common psychological symptom was agitation, occurring in more than 70% of cases. In most individuals the agitation was severe and accompanied by either aggression or abnormal behaviour. A fifth of individuals presented with acute low mood and suicidality which had not been present before mephedrone ingestion. Psychotic symptoms were present in 40% of individuals, with 88% of these reporting either daily or heavy mephedrone use for at least 4 weeks prior to presentation and 75% having had a prior history of mental illness. Psychiatric symptoms and psychological distress responded in a predictable manner to appropriate pharmacotherapy.
Discussion
This is the first known case series documenting the adverse psychological or psychiatric effects of mephedrone use. Before its reclassification, mephedrone was aggressively marketed by various online suppliers as a ‘legal high’ - a legal alternative to cocaine and amphetamine, which in the UK could also be purchased in specialised ‘head’ shops.
Case reports and case series are traditionally thought of as methodologically weak and rank low in the hierarchy of evidence. Recognised areas of weakness in this series are that all mephedrone use is self-reported, as there is no routine toxicology screening to confirm the presence of the drug. Currently, routine laboratory testing is not available for mephedrone in the National Health Service in Scotland, although testing of hair, saliva and urine for mephedrone has recently been developed by the private sector.
Another weakness is that in some cases there was little information available on the amount of mephedrone consumed and (heavy) usage was self-reported. It is also difficult to ascertain how common the occurrence of these adverse events is, given that no accurate rate of population use is available. Although we acknowledge the limitations of this paper, it is difficult to see how the psychological effects of mephedrone could easily be studied. Another problem is that there is little published literature on the pharmacokinetics or pharmacodynamics of mephedrone in animals or human volunteers and available information on the effects of mephedrone is limited to unconfirmed user reports. Furthermore, a person may believe they are using mephedrone as sold, but the precise purity, dose or even nature of the compound may not be transparent. It appears that the desired effects and common adverse effects are similar to those seen with other stimulant drugs such as 3,4-methylenedioxymethamphetamine (MDMA), amphetamines and cocaine. From information available to date it would appear that mephedrone not only is structurally similar to amphetamine, but appears to share common negative psychological side-effects with it, which may be more pronounced in those with a history of mental illness.
In this case series, of the eight patients presenting with florid psychotic symptoms following mephedrone ingestion, four had a history of psychotic illness and two had a history of depression. Two patients were taking antipsychotic medication at time of presentation. This finding would appear to follow a similar pattern to results which have been shown in previous studies looking at stimulant-induced psychotic symptoms. A systematic review in 2004 indicated that stimulant use could result in a short-lived psychotic reaction, more pronounced in those with pre-existing psychotic symptoms, and was unaffected by adherence to regular antipsychotic medication. Reference Curran, Byrappa and McBride8 Over 65% of our sample reported regular or heavy use of mephedrone prior to presentation, with 88% of the patients who presented with psychotic symptoms also reporting heavy use. Although data on dependence are currently limited to case reports which suggest that individuals can rapidly become regular users, with some reporting early cravings for mephedrone, Reference Wood, Davies, Puchnarewicz, Button, Archer and Ramsey9 the analogy to amphetamines would suggest that chronic mephedrone use is likely to carry a similar risk of dependency. 10 A report from the European Monitoring Centre for Drugs and Drug Addiction in 2010 commented that the more severe unwanted effects of mephedrone appear (anecdotally) to be associated with higher doses or prolonged use. 11
Suicides involving mephedrone were widely reported in the popular press, although few have been substantiated by toxicology results. In this case series one patient died by suicide following an acute increase in psychotic symptoms after ingesting mephedrone (self-reported ingestion), but it is hard to draw conclusions with regard to the suicide-inducing potential of mephedrone.
Despite the limitations of these and other related data, it is clear that mephedrone use can result in both physical and mental harm, similar in nature to that caused by other controlled stimulant drugs. The ACDM has been criticised for prematurely recommending that mephedrone be classified as a class B drug, Reference Nutt12 but our data would suggest that mephedrone use can have serious harmful consequences. However, market forces have meant that as soon as one substance is made illegal, protean alternatives are produced (‘Ivory Wave’ would be one example), which in turn lead to further presentations to emergency services. More work needs to be done in ascertaining the risks to health posed by mephedrone and by the so-called ‘legal highs’ - just because something is legal does not mean it is safe.
Acknowledgements
We thank Carole Haxton, the mental health assessment service, and the Department of Psychological Medicine, Royal Infirmary, Edinburgh.






eLetters
No eLetters have been published for this article.